
We begin with the elements in their most common states, Cs( s) and F 2( g). Hence, the valence of carbon is 4.\): The Born-Haber cycle shows the relative energies of each step involved in the formation of an ionic solid from the necessary elements in their reference states. The total iron-binding capacity (TIBC) of serum is a measure of total serum transferrin (apotransferrin, monotransferrin, diferric transferrin) concentration because insignificant amounts of plasma iron are bound to other proteins. For example, in methane \ molecules, carbon atoms form a total of four bonds with four hydrogen atoms. Ion-selective electrode sensors have been classified according to the membrane type and include the following electrode classifications: (1) homogeneous crystalline membrane electrodes (e.g. Note: The valency of an atom represents the number of bonds it can form with other atoms. A total iron binding capacity (TIBC) test is a type of blood test that gauges whether there’s too much or too little of the mineral in your bloodstream. So the combining capacity of an element is known as its valency. It provides a measure of how difficult it is to extend a material, with a value given by the ratio of tensile strength to. It is often used for inorganic anions (e.g., chloride, nitrate, and sulfate) and inorganic cations (e.g., lithium, sodium, and potassium). 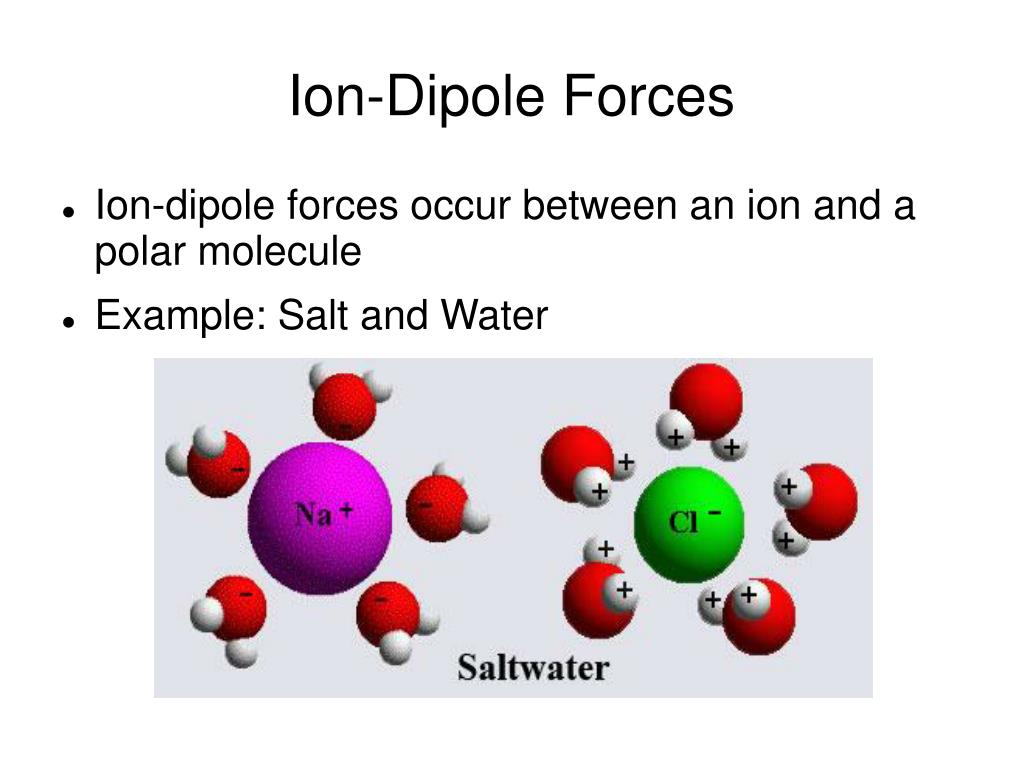
This technique is used to analyze ionic substances. A measure of the stiffness of a substance. A second sub-category of liquid chromatography is known as ion-exchange chromatography. To attain stability, magnesium loses two valence electrons and completes its octet. Specific heat capacity (J kg 1 K 1) Specific heat capacity is the amount of energy needed to change the temperature of a kilogram of a substance by 1 K. Provide proper financial statements and reports Work with a construction CPA so you know which reports the surety will be looking at, and make sure they are presented professionally. On the other hand, magnesium has two electrons. There are four primary ways for contractors to boost their bonding capacity. To attain stability, sodium loses one valence electron and completes its octet. For example, sodium has only one electron in its valence shell. In this way, it attains stability.įor metals, you can calculate the valence from its valence shell electronic configuration. In an effort to arrive at the correct answer, we have thoroughly scrutinized each option and taken into account all relevant information that. Upon examining the given clues, we have managed to identify a total of 1 possible solutions for the crossword clue Ion bonding capacity measure. To attain stability, oxygen gains two valence electrons and completes its octet. Ion bonding capacity measure Crossword Clue. Similarly, oxygen has 6 valence electrons. To attain stability, chlorine gains one valence electron and completes its octet. The outermost shell is also called the valence shell. Lewis Structures Atoms react with one another to form molecules in order to achieve a more stable electronic configuration The valence electrons are those. 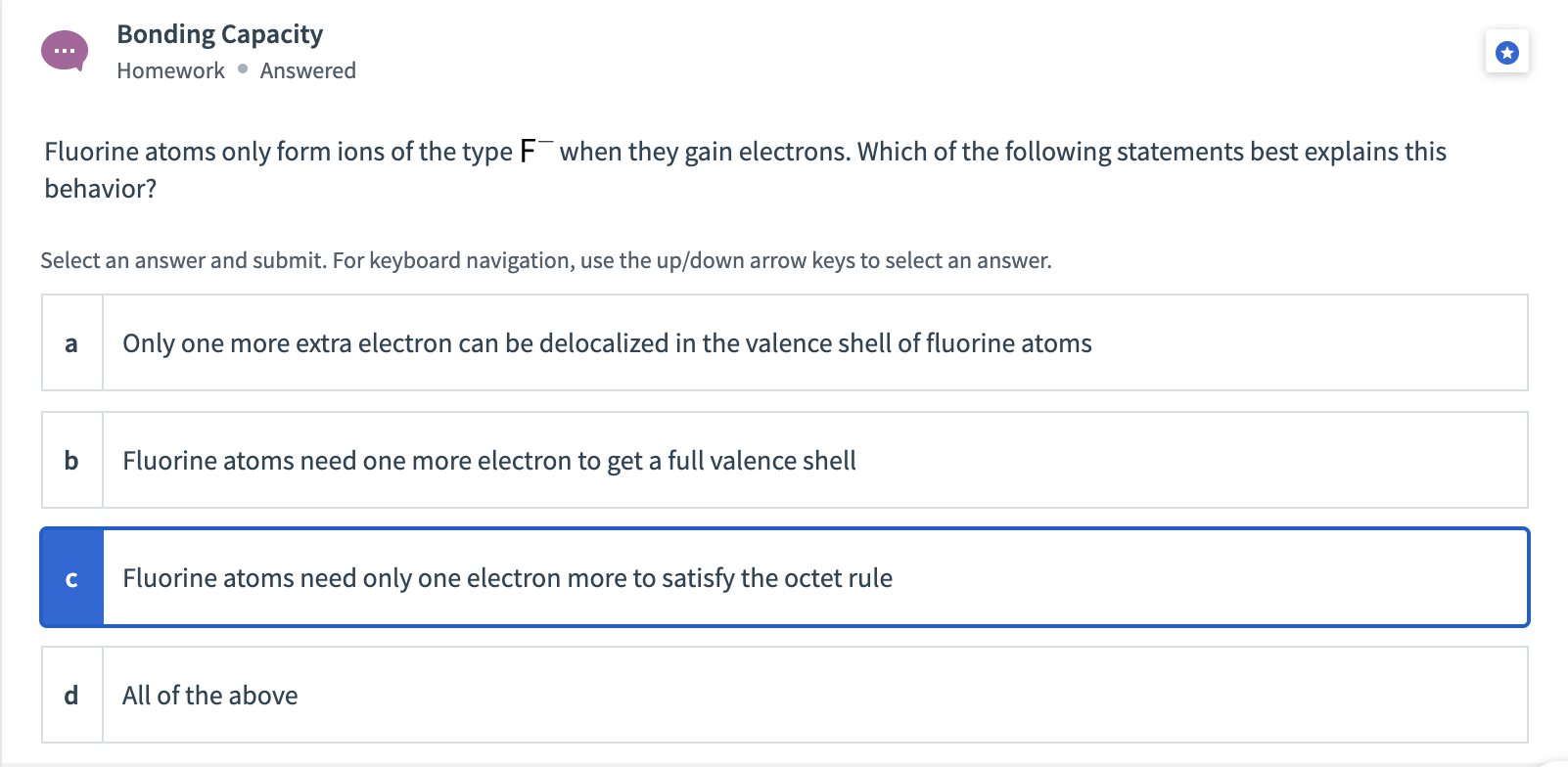
A measure of the stiffness of a substance. The valence electrons are the number of electrons present in the outermost shell. Specific heat capacity (J kg 1 K 1) Specific heat capacity is the amount of energy needed to change the temperature of a kilogram of a substance by 1 K. (WHC) (or water-binding capacity, or water-absorption capacity) is a measure of. calorimeter: a device used to measure the heat of a chemical reaction. CHEMICAL AND PHYSICAL CHARACTERISTICS OF MEAT Water-Holding Capacity. The combining capacity of an element is known as its valency.įor a nonmetal, you can calculate its valence by subtracting the number of valence electrons from 8. A single bond, double bond, and triple bond all count as a single bonding group of. The combining capacity of an element is dependent upon the number of electrons needed by an atom to achieve stable electronic configuration. Thus, if you count the number of bonds around the central atom, you can determine its combining capacity. Binding affinity is typically measured and reported by the equilibrium dissociation constant (KD), which is used to evaluate and rank order strengths of. 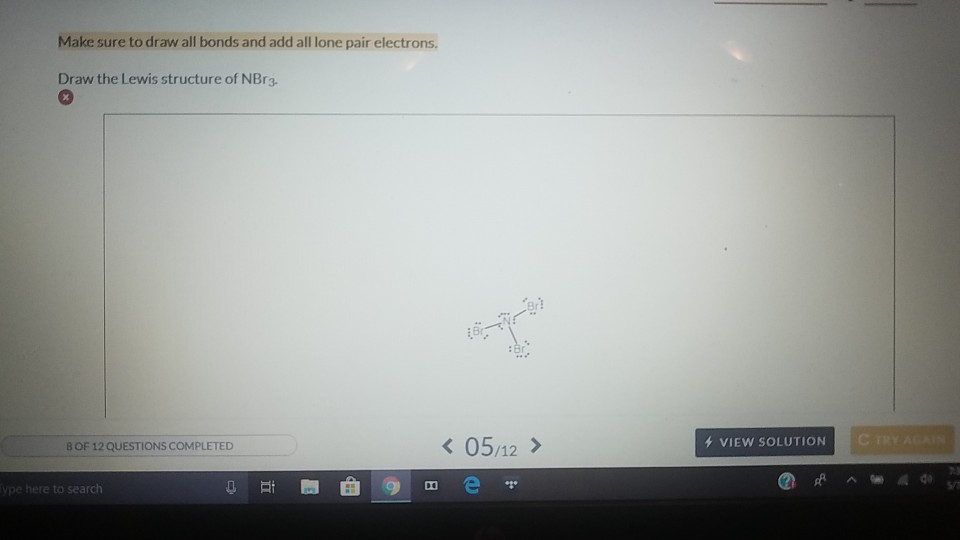
Hint: The combining capacity of an element will tell you the number of atoms that are bonded to the given atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
